Zhang lab @ MIT & Broad
Biological Discovery & Engineering
Image credit to Allison Colorado (2024)
We explore and study biological diversity to understand nature and discover systems and processes that may be harnessed through bioengineering for the improvement of human well-being.
Recent Highlights
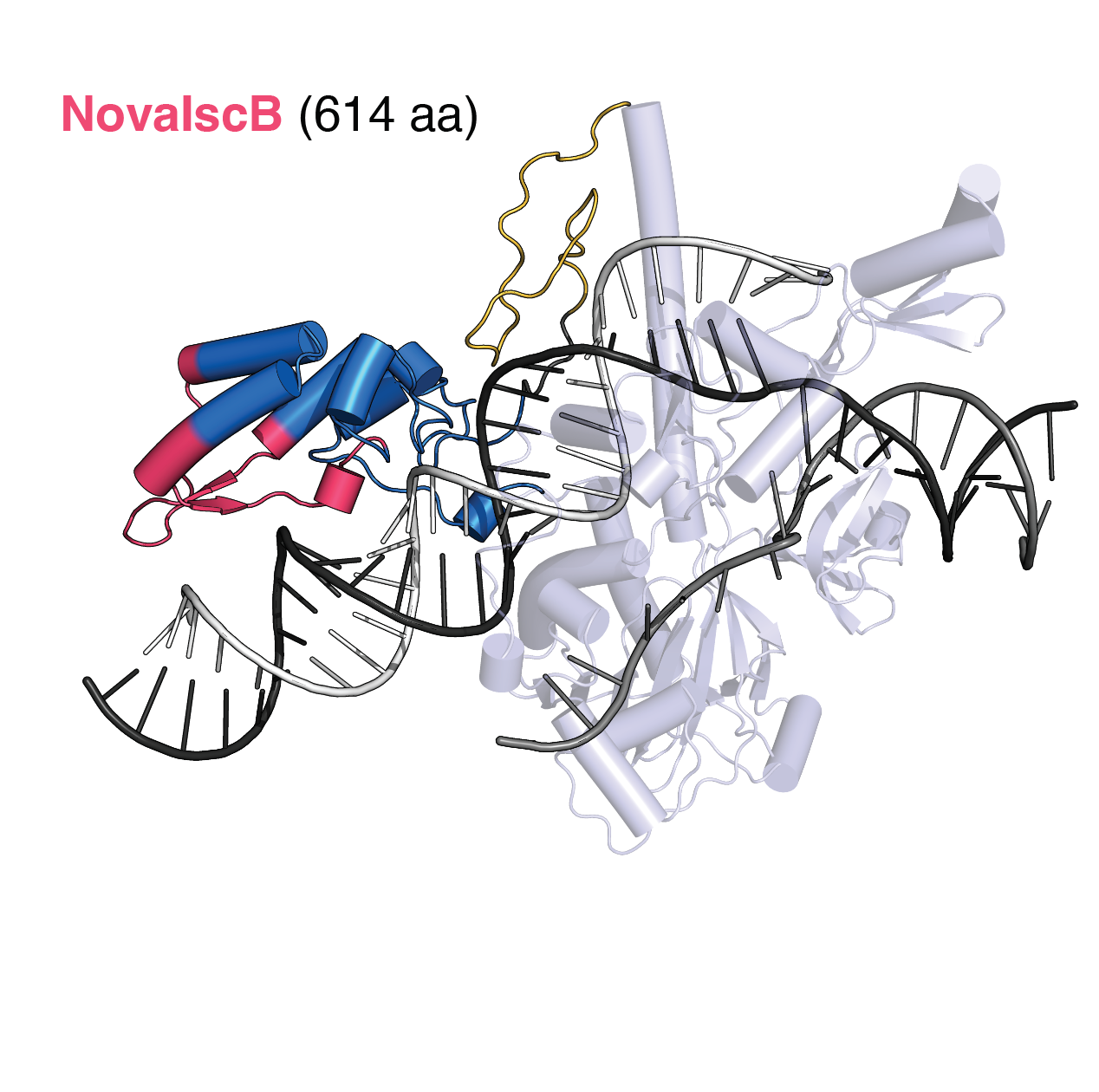
Evolution-guided protein design of IscB for persistent epigenome editing in vivo
May 7, 2025
Kannan, Altae-Tran, Zhu et al. combined evolution- and structure-guided engineering to generate NovaIscB, a compact OMEGA editor and adapt it for gene repression. Read more in Nature Biotechnology.
An an all-RNA system for DNA insertion with R2 retrotransposons
July 2, 2025
Edmonds et al. demonstrate the R2 retrotransposon from zebra finch is active in human cells and can be engineered for efficient transgene insertion. Read more in Nature Communications.
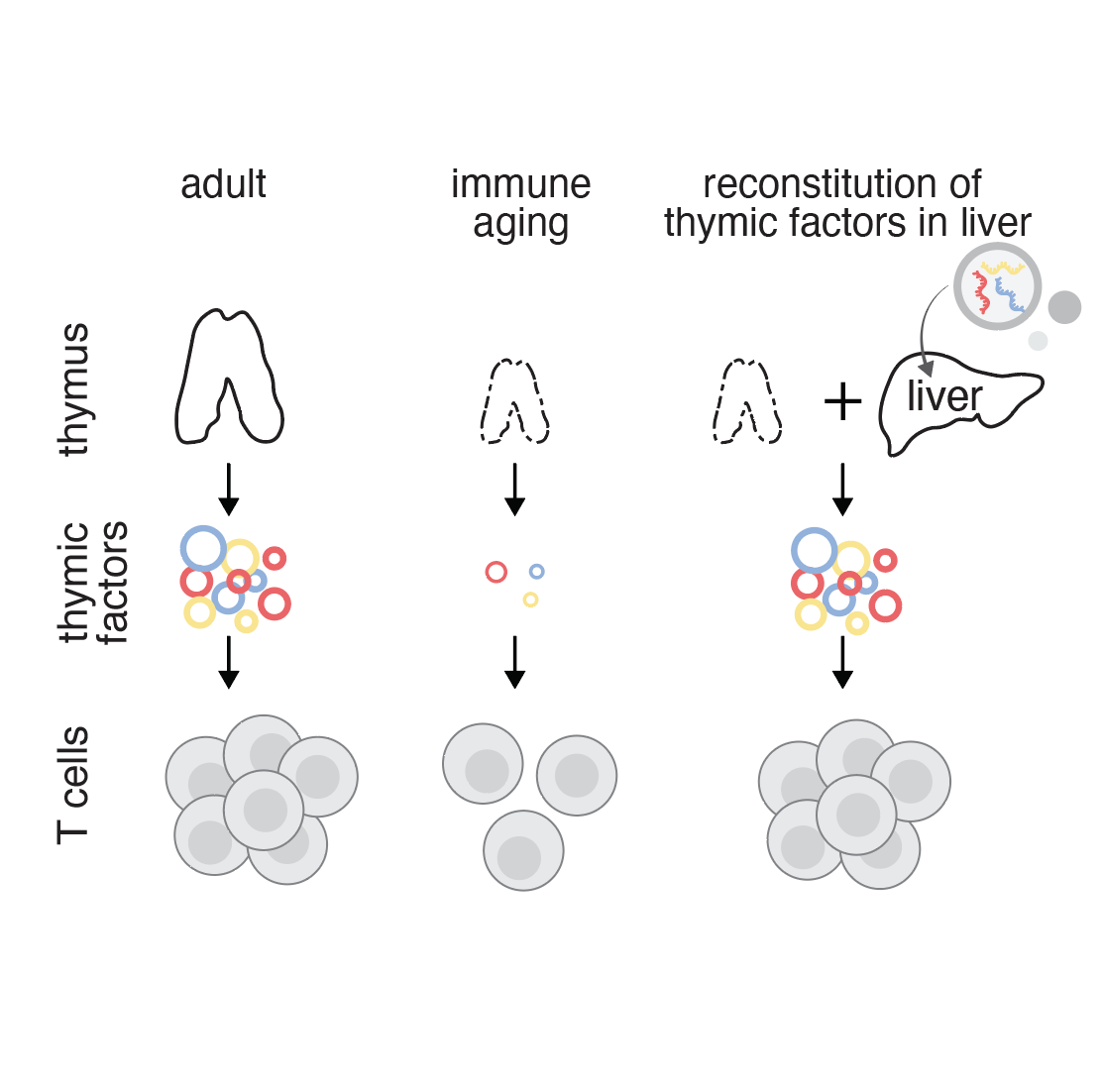
Transient hepatic reconstitution of trophic factors enhances aged immunity
December 17, 2025
Friedrich et al. show that the aged immune system can be transiently boosted by expressing a cocktail of three trophic factors in the liver. Read more in Nature.